- Rx Products
- Derma-Smoothe/FS® Scalp Oil
- Derma-Smoothe/FS® Body Oil
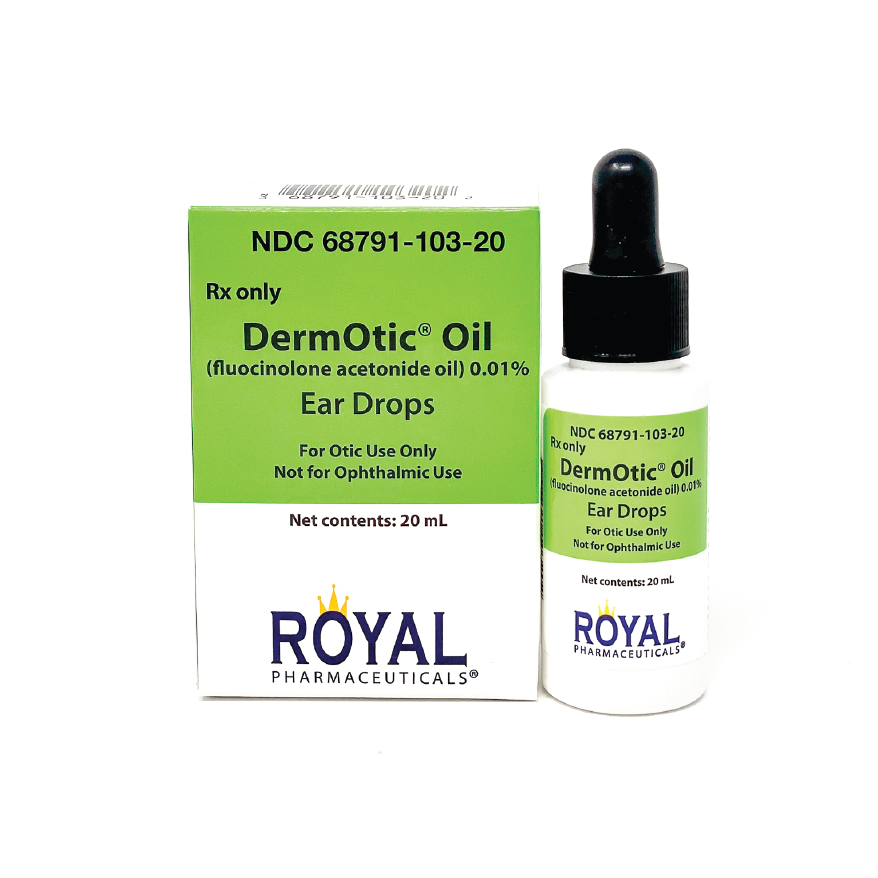
- DermOtic® Oil Ear Drops
- Tolak®
- Canada – PrDerma-Smoothe/FS® Body Oil
- Canada – PrDerma-Smoothe/FS® pour le corps
- Canada – Pr Derma-Smoothe/FS® Scalp Oil
- Canada – PrDerma-Smoothe/FS® pour le cuir chevelu
- Canada – PrDermOtic® Oil Ear Drops
- Canada – PrGouttes otiques Huile DermOtic®
- Canada – Pr Tolak® (fluorouracil) Cream 4%
- Canada – PrTolak®Crème (fluoro-uracile) à 4%
- News
- About Us
- Contact Us